
Making the structure of ‘fire ice’ with nanoparticles
The structure harnesses a strange physical phenomenon and could enable engineers to manipulate light in new ways.

The structure harnesses a strange physical phenomenon and could enable engineers to manipulate light in new ways.
Written by Kate McAlpine and originally published by Michigan Engineering.
Cage structures made with nanoparticles could be a route toward making organized nanostructures with mixed materials, and researchers at the University of Michigan have shown how to achieve this through computer simulations.
The finding could open new avenues for photonic materials that manipulate light in ways that natural crystals can’t. It also showcased an unusual effect that the team is calling entropy compartmentalization.
“We are developing new ways to structure matter across scales, discovering the possibilities and what forces we can use,” said Sharon Glotzer, the Anthony C. Lembke Department Chair of Chemical Engineering, who led the study published today in Nature Chemistry. “Entropic forces can stabilize even more complex crystals than we thought.”
While entropy is often explained as disorder in a system, it more accurately reflects the system’s tendency to maximize its possible states. Often, this ends up as disorder in the colloquial sense. Oxygen molecules don’t huddle together in a corner—they spread out to fill a room. But if you put them in the right size box, they will naturally order themselves into a recognizable structure.
Nanoparticles do the same thing. Previously, Glotzer’s team had shown that bipyramid particles—like two short, three-sided pyramids stuck together at their bases— will form structures resembling that of fire ice if you put them into a sufficiently small box. Fire ice is made of water molecules that form cages around methane, and it can burn and melt at the same time. This substance is found in abundance under the ocean floor and is an example of a clathrate. Clathrate structures are under investigation for a range of applications, such as trapping and removing carbon dioxide from the atmosphere.

Unlike water clathrates, earlier nanoparticle clathrate structures had no gaps to fill with other materials that might provide new and interesting possibilities for altering the structure’s properties. The team wanted to change that.
“This time, we investigated what happens if we change the shape of the particle. We reasoned that if we truncate the particle a little, it would create space in the cage made by the bipyramid particles,” said Sangmin Lee, a recent doctoral graduate in chemical engineering and first author of the paper.
He took the three central corners off each bipyramid and discovered the sweet spot where spaces appeared in the structure but the sides of the pyramids were still intact enough that they didn’t start organizing in a different way. The spaces filled in with more truncated bipyramids when they were the only particle in the system. When a second shape was added, that shape became the trapped guest particle.
Glotzer has ideas for how to create selectively sticky sides that would enable different materials to act as cage and guest particles, but in this case, there was no glue holding the bipyramids together. Instead, the structure was completely stabilized by entropy.
“What’s really fascinating, looking at the simulations, is that the host network is almost frozen. The host particles move, but they all move together like a single, rigid object, which is exactly what happens with water clathrates,” Glotzer said. “But the guest particles are spinning around like crazy—like the system dumped all the entropy into the guest particles.”
This was the system with the most degrees of freedom that the truncated bipyramids could build in a limited space, but nearly all the freedom belonged to the guest particles. Methane in water clathrates rotates too, the researchers say. What’s more, when they removed the guest particles, the structure threw bipyramids that had been part of the networked cage structure into the cage interiors—it was more important to have spinning particles available to maximize the entropy than to have complete cages.
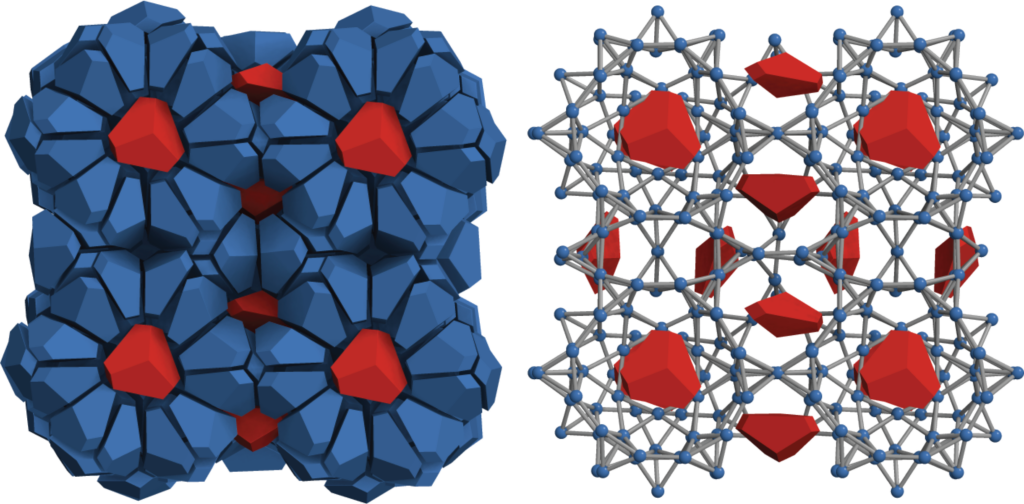
“Entropy compartmentalization. Isn’t that cool? I bet that happens in other systems too—not just clathrates,” Glotzer said.
Thi Vo, a former postdoctoral researcher in chemical engineering at U-M and now an assistant professor of chemical and biomolecular engineering at the Johns Hopkins University, contributed to the study.
This study was funded by the Department of Energy and Office of Naval Research, with computing resources provided by the National Science Foundation’s Extreme Science and Engineering Discovery Environment and the University of Michigan.
Glotzer is also the John Werner Cahn Distinguished University Professor of Engineering, the Stuart W. Churchill Collegiate Professor of Chemical Engineering, and a professor of materials science and engineering, macromolecular science and engineering, and physics.